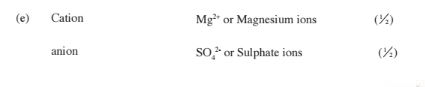2014 KCSE Chemistry Past Paper
3.6.1 Chemistry Paper 1 (233/1)
1 Explain how the hotness of a Bunsen burner flame can be increased. (1 mark)
2 When dilute hydrochloric acid was reacted with solid B, a colourless gas which extinguished a buming splint was produced. When an aqueous solution of solid B was tested with a blue litmus paper, the paper turned red/ pink.
(a) Identify the anion present in solid B. (1 mark)
(b) Write an ionic equation for the reaction between solid B and dilute hydrochloric acid. (1 mark)
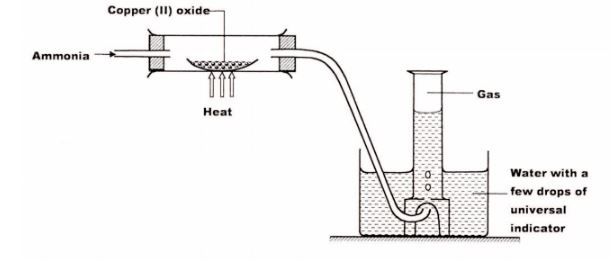
3 Dry ammonia gas was passed over heated lead (II) oxide and the products passed over anhydrous copper (II) sulphate as shown in the diagram below.
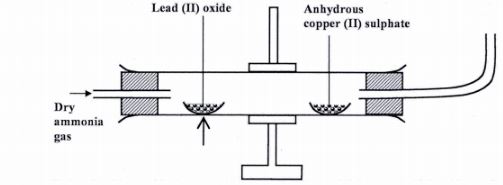
State:
(a) two observations made in the combustion tube. (2 marks)
(b) the property of ammonia gas shown in this experiment. (1 mark)
4 Starting with zinc sulphate solution, describe how a sample of Zinc oxide can be obtained. (3 marks)
5 Explain how conduction of electricity takes place in the following:
(a) iron metal; (1 mark)
(b) molten lead (ll) iodide. (1 mark)
6 100 cm3 of a sample of ethane gas diffuses through a porous pot in 100 seconds. What is the molecular mass of gas Q if 100 cm} of the gas diffuses through the same porous pot in l2l seconds under the same conditions?
(C = 12.0, H = 1.0) (3 marks)
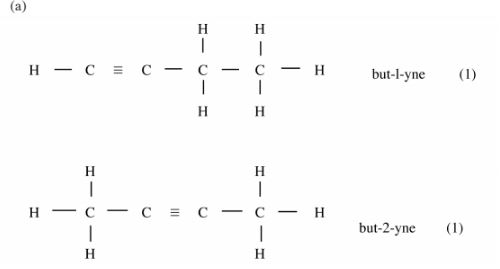
7 (a) Draw and name the isomers of butyne. (2 marks)
(b) State one use of polystyrene. (1 mark)
8 Complete the nuclear reaction below:
imageee (1 mark) (b) State two uses of radioisotopes in health. (2 marks)
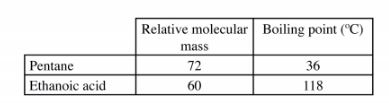
Explain the large difference in boiling point between ethanoic acid and pentane. (2 marks) 10 One of the ores of copper has the formula, CuFeS2.
(a) Describe how iron in the ore is removed during extraction of copper metal. (1 mark)
(b) State two environmental problems associated with extraction of copper metal. (2 marks)
ll Study the flow chart below and answer the questions that follow.
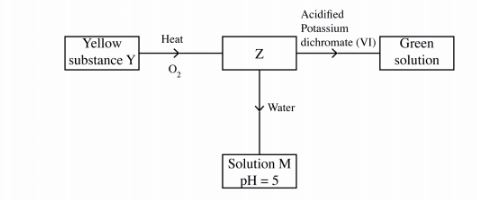
Identify Z and M. (2 marks)
Z …………………………………………………. ..
M …………………………………………………….
12 The table below shows the pH values of solutions A, B, C and D.
Solution
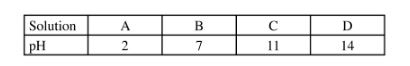
Select solutions in which a sample of lead (ll) hydroxide is likely to dissolve. Give reasons for each solution selected. (3 marks)
13 100 cm3 of 0.05 M sulphuric (VI) acid were placed in a flask and a small quantity of anhydrous sodium carbonate added. The mixture was boiled to expel all the carbon (IV) oxide. 25 cmi of the resulting solution required 18 cm3 of 0.l M sodium hydroxide solution to neutralise it. Calculate the mass of sodium carbonate added. (Na : 23.0; O : 16.0; C = 12.0) (3 marks)
14 When 20 cm3 of l M sodium hydroxide was mixed with 20 cm3 of 1 M hydrochloric acid, the temperature rose by 6.7 “C. Assuming the density of the solution is l g/cm3 and the specific heat capacity of the solution is 4.2 J g” k”;
(a) calculate the molar heat of neutralisation; (2 marks)
(b) when the experiment was repeated with 1 M ethanoic acid, the temperature change was found to be lower than that with 1 M hydrochloric acid. Explain. (1 mark)
15 Study the set-up below and answer the questions that follow.
(a) Write an equation for the reaction between ammonia and copper (II) oxide. (1 mark)
(b) During the experiment, the colour of the contents in the water trough changed. State the colour change observed and give an explanation. (2 marks)
16 A measuring cylinder fitted with moist steel wool was inverted in a trough of water as shown in the diagram below.
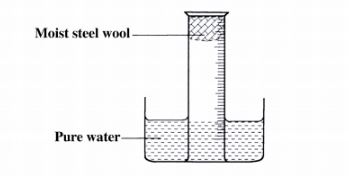
(a) State and explain the observations made on the:
(i) moist steel wool after four days. (1 mark)
(ii) water level in the measuring cylinder after four days. (1 mark)
(b) What would be the effect of using steel wool moistened with salty water? (1 mark)
17 In an experiment on rates of reaction, potassium carbonate was reacted with dilute sulphuric
(VI) acid.
(a) What would be the effect of an increase in the concentration of the acid on the rate of the reaction? (1 mark)
(b) Explain Why the rate of reaction is found to increase with temperature. (2 marks) 18 Use the part of the periodic table given below to answer the questions that follow.
(Letters are not the actual symbols of the elements).
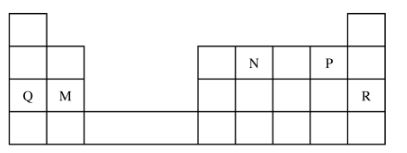
(a) Identify the element that forms giant covalent structures. (1 mark)
(b) Identify one element that does not form compounds. (1 mark)
(c) Write the formula for the nitride of M. (1 mark)
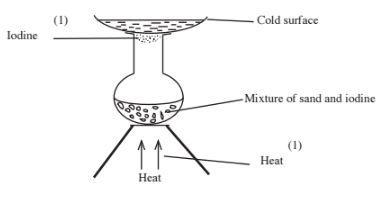
19 Draw a set up that can be used to separate a mixture of sand and iodine. (3 marks)
20 In the contact process, during the production of sulphur (VI) oxide, a catalyst is used.
Give two reasons why vanadium (V) oxide is preferred to platinum. (2 marks)
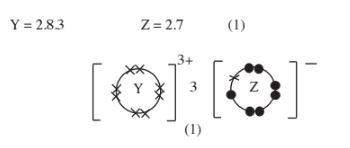
21 Given that the atomic number of Y is 13 and that of Z is 9:
(a) write the electronic arrangement of Y and Z; (1 mark)
(b) draw the dot ( .) and cross (X) diagram for the compound fomied by Y and Z. (1 mark)
22 The set up below was used to separate a mixture of methanol and propanol. Study it and answer the questions that follow.
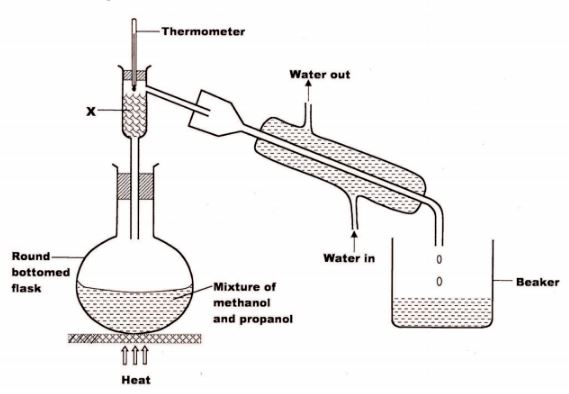
(a) State the function of X.(1 mark)
(b) Which liquid will collect first in the beaker? Give a reason.(2 marks)
23 Study the flow chart below and answer the questions that follow.
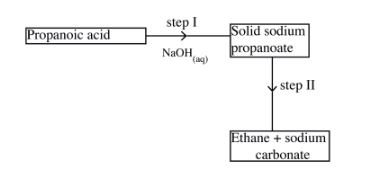
(a) Name the process in step l.(1 mark)
(b) Identify the reagent in step II.(1 mark)
(c) Give one use of ethane.(1 mark)
24 (a) A student electrolysed dilute sodium chloride solution using inert carbon electrodes. Name the products at:
(i) anode;
(ii) cathode.
(2 marks) (b) If the experiment was repeated using concentrated sodium chloride instead of dilute sodium chloride solution, Write the half equation at the anode. (1 mark)
25 An organic compound had the following composition 37.21% carbon, 7.75% hydrogen and the rest chlorine. Detemiine the molecular formula of the compound, given that the molecular mass of the compound is 65. (C : 12.0; H : 1.0; Cl : 35.5). (3 marks)
26 Cotton is a natural polymer. State one advantage and one disadvantage of this polymer. (2 marks)
Advantage: …………………………………………….. ..
Disadvantage: …………………………………………… ..
27 (a) Name a suitable solvent for extracting an indicator from flowers; (1 mark)
(b) Give a reason Why the solvent named in (a) above is used. (1 mark)
28 A student used the set up below to prepare a sample of nitrogen gas.
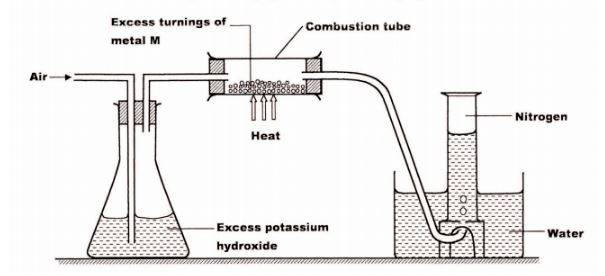
(a) State the function of potassium hydroxide in the set up. (1 mark)
(b) Give a suitable metal M for use in the combustion tube.
(c) Give a reason why the nitrogen gas obtained is not pure. (1 mark)
29 (a) What is meant by the term radical? (1 mark)
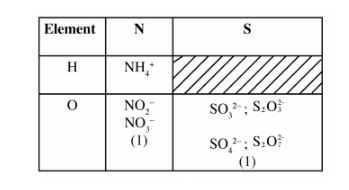
(b) The table below contains atoms that form common radicals. Complete the table to show radicals formed from various atoms.
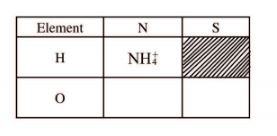
(2 marks)
30 A gas jar full of chlorine water was inverted over water and allowed to stand for sometime.
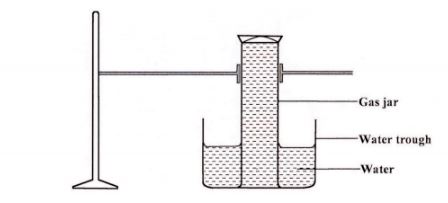
(a) State and explain two observations made in the gas jar after some time. (2 marks)
(b) Write the equation for the reaction between chlorine and hot concentrated potassium hydroxide. (1 mark)
3.6.2 Chemistry Paper 2 (233/2)
1 (a) The set-up below was used to prepare dry hydrogen chloride gas, and investigate its effect on heated iron filings.
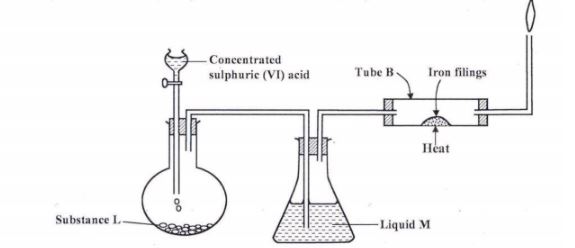
(i) Name substance L (1 mark)
(ii) Name liquid M. (1 mark)
(m) What will be observed in tube B (1 mark)
(iv) Write an equation for the reaction that occurs in tube B. (1 mark)
(v) Why is the gas from tube B burnt? (1 mark)
(b) (i) Explain the following observations:
(I) a white precipitate is formed when hydrogen chloride gas is passed through aqueous silver nitrate. (1 mark)
(II) hydrogen chloride gas fumes in ammonia gas. (1 mark)
(ii) State two uses of hydrogen chloride gas. (1 mark)
(c) The diagram below is a representation of an industrial process for the manufacture of a bleaching powder.
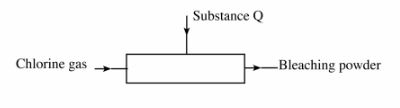
(i) Name substance Q. (1 mark)
(ii) When the bleaching powder is added to water during washing, a lot of soap is used. Explain. (1 mark)
2 (a) The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbols of the elements.
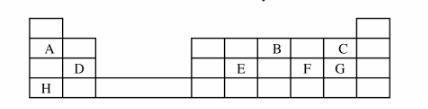
Select the most reactive metal. Explain. (2 marks)
(ii) Select an element that can form an ion with a charge of 3 . (1 mark)
(iii) Select an alkaline earth metal. (1 mark)
(iv) Which group l element has the highest first ionization energy? Explain. (2 marks)
(v) Element A combines with chlorine to form a chloride of A. State the most likely pH value of a solution of a chloride of A. Explain. (2 marks)
(b) (i) Explain why molten calcium chloride and magnesium chloride conduct electricity While carbon tetrachloride and silicon tetrachloride do not. (2 marks)
(ii) Under the same conditions, gaseous neon was found to diffuse faster than gaseous fluorine. Explain this observation. (F = 19.0; Ne = 20.0) (2 marks)
3 (a) Draw the structures of the following:
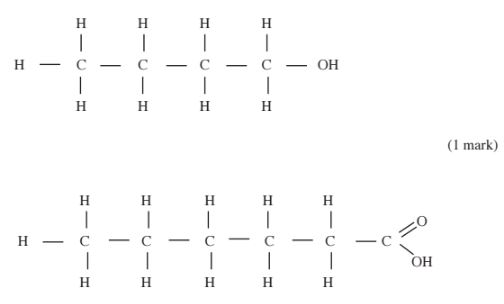
(i) Butan- l -ol (1 mark)
(ii) Hexanoic acid. (1 mark)
(b) Study the flow chart below and answer the questions that follow.
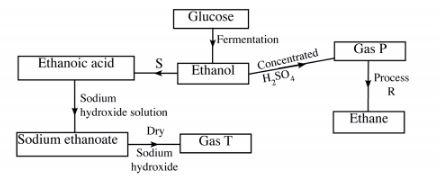
(i) State the conditions necessary for fermentation of glucose to take place. (1 mark)
(ii) State one reagent that can be used to carry out process S. (1 mark)
(m) Identify gases: (2 marks)
P: ………………………………… ..
T: ………………………………… ..
(iv) How is sodium hydroxide kept dry during the reaction? (1 mark)
(v) Give one commercial use of process R. (1 mark)
(c) When one mole of ethanol is completely burnt in air, l370kj of heat energy is released. Given that 1 litre of ethanol is 780g, calculate the amount of heat energy released when l litre of ethanol is completely burnt. (3 marks)
(C = 12.0; H= 1.0; O = 16.0)
(d) State two uses of ethanol other than as an alcoholic drink. (2 marks) 4 (a) Other than temperature, state two factors that determine the rate of a chemical reaction. (1 mark)
(b) A solution of hydrogen peroxide was allowed to decompose and the oxygen gas given off collected. After 5 minutes, substance G was added to the solution of hydrogen peroxide. The total volume of oxygen evolved was plotted against time as shown in the graph below.
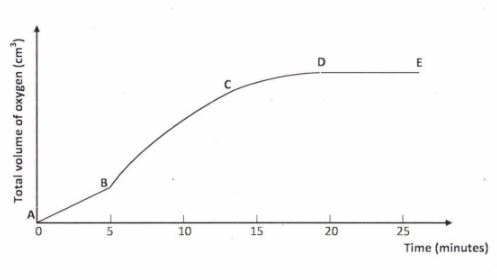
(i) Describe the procedure of determining the rate of the reaction at minute l2. Explain. (3 marks)
(ii) How does the production of oxygen in region AB compare with that in region BC? (2 marks)
(iii) Write an equation to show the decomposition of hydrogen peroxide. (1 mark)
(c) Sulphur (IV) oxide reacts with oxygen to form Sulphur (V1) oxide as shown in the equation below:

(i) Explain the effect on the yield of SO3 of lowering the temperature for this reaction . (2 marks)
(ii) Name one catalyst used for the reaction. (1 mark)
5 (a) The scheme below shows some of the reactions of solution D. Study it and answer the questions that follow.
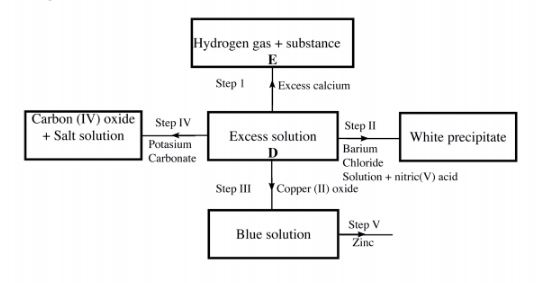
(i) Give a possible cation present in solution D. (1 mark)
(ii) Write an ionic equation for the reaction in Step II. (1 mark)
(iii) What observations would be made in Step V? Give a reason. (2 marks)
(iv) Explain why the total volume of hydrogen gas produced in step 1 was found to be very low although calcium and solution D were in excess. (2 marks)
(v) State one use of substance E. (1 mark)
(b) Starting with solid sodium chloride, describe how a pure sample of lead (ll) chloride can be prepared in the laboratory. (3 marks)
(c) (i) State a property of anhydrous calcium chloride which makes it suitable for use as a drying agent for chlorine gas. (1 mark)
(ii) Name another substance that can be used to dry chlorine gas. (1 mark)
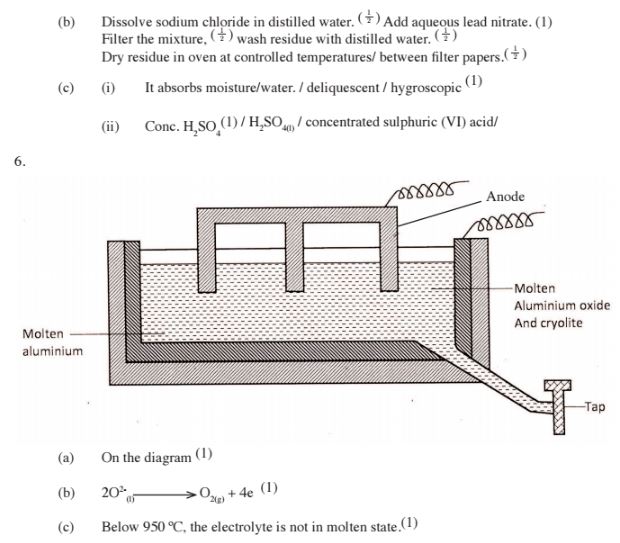
6. The diagram below represents a set up of an electrolyte cell that can be used in the production of aluminium
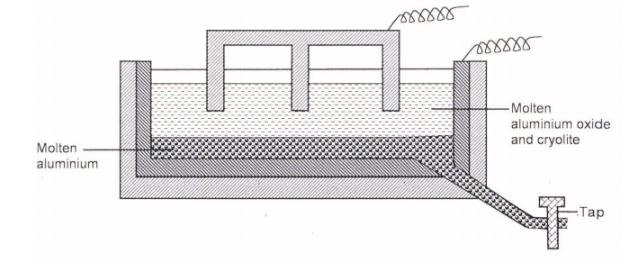
(a) On the diagram, label the anode (1 mark)
(b) Write the equation for the reaction at the anode (1 mark)
(c)Give a reason why the electrolyte process is not carried out below 950°C (1 mark)
(d) Give a reason why the production of aluminium is not carried out using reduction process (1 mark)
(e) Give two reasons why only the aluminium ions are discharged (2 marks)
(f) State two properties of duralumin that makes it suitable for use in aircraft industry (g)Name two environmental effects caused by extraction of aluminum (2 marks)
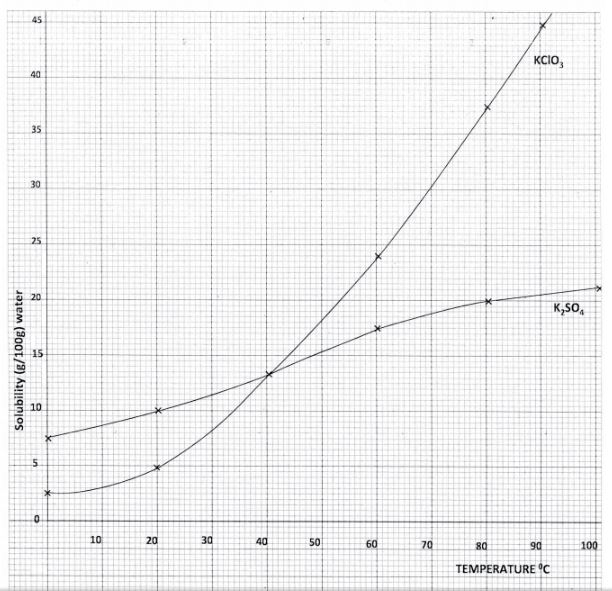
7. (a) Dissolving of potassium nitrate tn water rs an endothermic process Explain the effect of increase in temperature on the solubility of potassium nitrate (b) The table below shows the solubility of potassium sulphate and potassium chlorate (V) at different temperatures.
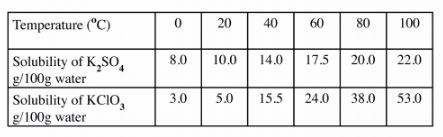
(i) Draw the solubility curves for both salts on the same axis. (Temperature on the X-axis). (3 marks)
(ii) A solution of potassium sulphate contains 20g of the salt dissolved in 100g of water at 100°C. This solution is allowed to cool to 25°C. I At what temperature will crystals first appear? (1 mark)
ll What mass of crystals will be present at 25“C? (1 mark)
(111) Which of the two salts is more soluble at 30°C? (1 mark)
(iv) Determine the concentration of potassium sulphate in moles per litre when the solubility of the two salts a.re the same (K = 39.0, O = l6.0; S = 32.0). (3 marks) (v) 100g of water at 100°C contains 19g of potassium sulphate and 19g of potassium chlorate (V). Describe how a solid sample of potassium sulphate at 60 C can be obtained. (2 marks)
3.6.3 Chemistry Paper 3 (233/3)
1 You are provided with:
– solution J containing copper (ll) ions.
solution K, 0.1 M sodium thiosulphate.
aqueous potassium iodide, solution L.
solution N, sodium hydroxide.
starch indicator, solution M.
You are required to determine the:
– concentration of copper (ll) ions in solution J;
– enthalpy change of reaction between copper (ll) ions and hydroxide ions.
PROCEDURE 1
(a) Using a pipette and pipette filler, place 25.0 cm-‘ of solution J in a 250 ml volumetric flask. Add distilled water to make up to the mark. Label this as solution J2. Retain solution J for use in procedure II.
Place solution K in a burette. Using a clean pipette and pipette filler, place 25.0 cm3 of solution J2 in a 250 ml conical flask. Add 10 cm3 of potassium iodide, solution L.
Shake well, then add 2 cm3 of starch indicator, solution M. Titrate until a blue-black colour appears and continue titrating until the blue-black colour just disappears.
Record your readings in Table 1 below.
(c) Repeat step (b) two more times and complete Table 1.
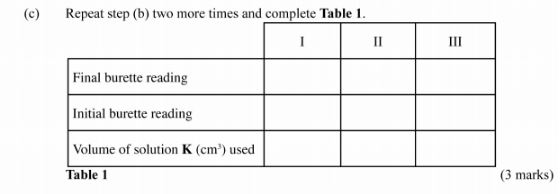
Calculate the;
(i) average volume of solution K used; (1 mark)
(ii) moles of sodium thiosulphate used; (2 marks)
(iii) concentration in moles per litre of copper (ll) ions in solution J given that the number of moles of copper (ll) ions in 25.0 cm} of solution J2 are the same as the moles of sodium thiosulphate used. (2 marks)
PROCEDURE 11
(a) Using a clean burette, place 5.0 cm3 of solution N into each of six (6) test-tubes. (b) Using a 100 ml measuring cylinder, place 20 cm3 of solution J in a 100 ml plastic beaker. Measure the temperature of solution J and record it in Table 2 below.
(c) To solution J in the beaker, add sodium hydroxide, solution N from one of the test-tubes. Stir the mixture with the thermometer and record in Table 2, the maximum temperature reached. Continue with step (d) IMMEDIATELY.
(d)Add the sodium hydroxide, solution N from another test-tube to the mixture obtained in (c) above, stir and record the maximum temperature reached in Table 2. Continue adding the sodium hydroxide, solution N from each of the other four test-tubes, stirring the mixture and recording the maximum temperature each time and complete Table 2.
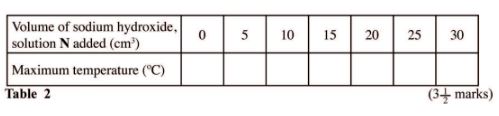
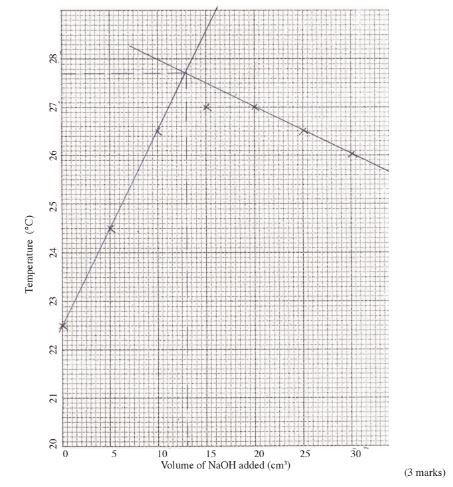
(i) On the grid provided, plot a graph of temperature (vertical axis) against volume of sodium hydroxide solution N added. (3 marks)

(ii) Using the graph, determine the:
I volume of sodium hydroxide, solution N that reacted completely with 20 cm3 of solution J; (2 marks)
II temperature change, AT, for the reaction; (1 mark)
(iii) enthalpy change of the reaction per mole of copper (ll)ions. (3 marks)
(Heat capacity I 4.2 J g”k”, density of the mixture I 1.0 g cm’3). 2 You are provided With substance P. Can”y out the tests below and Write your observations and inferences in the spaces provided.
(a) Describe the appearance of substance P. (1 mark)
(b) Place about one-third of substance P in a dry test-tube and heat it strongly.
(c) Place the remaining amount of substance P in a boiling tube. Add about 10 cm3 of distilled water and shake well. Retain the mixture for tests in (d) below.

(d) Use about 2 cml portions of the mixture obtained in (c) for tests (i) to (iii) below.
(i) Add two to three drops of aqueous barium nitrate to the mixture.

(ii) Add five drop of dilute nitric(V) acid to the mixture

(iii)Add to the mixture aqueous sodium hydroxide dropwise until in excess

(e) Give the formula of the cation and anion present in substance P
Cation: ………… ..
Anion: ………(1 mark)
3 You are provided with an organic substance Q. Carry out the following tests and record your observations and inferences in the spaces provided.
(a) Place about one-third of substance Q on a metallic spatula and ignite it with a Bunsen burner flame.

(b) Place the remaining amount of substance Q in a boiling tube and add about 10 cm} of distilled water. Heat the mixture and allow it to boil for about 30 seconds. Divide the mixture while still hot into two portions.
(i) To the first portion, add solid sodium hydrogen carbonate provided.

(ii) To the second portion, add two or three drops of acidified potassium manganate

2014 KCSE Chemistry Past Paper-Marking Scheme/Answers
4.6.1 Chemistry Paper 1 (233/1)
1. Increasing the size of the air hole/increase the amount of air/open air holes competely. (1)
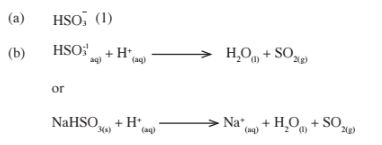
3. (a) – The anhydrous copper (II) Sulphate turns from White to blue. (1)
– A grey solid is formed/droplets of a colourless liquid condense at cool pal“t.(l)
(b) Reducing property.(l)
4. ~ Add soluble carbonate/Add soluble hydroxide. (1)
– Filter out the zinc carbonate/filter the zinc hydroxide. (1)
~ Heat strongly the ZnCO3 to decompose it to form ZnO/Heat strongly the Zn(OH)2 to decompose it to form ZnO. (1)
OR
– Heat to evaporate the water. (1)
– Heat ZnSO4 solid to decompose (1) to form ZnO/yellow solid. (1)
5. (a) delocalised electrons. (1)
(b) ions in the melt. (1)
6.

7. (a)
(b) Used in packaging – cushions electronics in boxes/insulation/models/ceiling strips/ crates or binding. (1)
8.
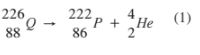
(b) (i) Cobalt 60 is used to detect the activity of the thyroid gland. (1)
(ii) To sterise equipment/treatment of cancer/radio active Na for disorders in blood circulation/Barium meal for ulcers/detect fractures in bones. (l)
9. The molecules of ethanoic acid interact through strong hydrogen bonding (1) forming a dimer while molecules of pentane have weak van der waals forces. (1) NB/ Ethanoic acid has hydrogen bonds while pentane does not have.
10. (a) Roast ore in air/heat in air. (l)

(b) – Acid rain that corrodes stone Work on buildings/land gulleys/dust pollution. (1)
– SO2 when breathed in causes bronchitis/chlorosis in plants. (1)
11. Z is SO2/ sulphur (IV) oxide. (1)
M is H2SO3/ sulphuric (IV) acid. (1)
12. A(l)andD(l)
A is acidic it Will neutralise Pb(OH)ZW to form salt and Water, (%)
D is a strong base it Will react with Pb (OH)2W to form a complex ion. (72)
Lead (II) hydroxide is amphoteric.
13.

14.

(b) Some energy is used to ionise the weak acid first before it can neutralise. So not all energy is used in neutralisation. (l)
15.
(b) It changed purple (1)
The excess ammonia makes solution basic which turns purple with universal indicator. (1)
16. (a) (i) It turned brown /blue/violet/green. (1)
(ii) The Water level rose up the gas jar/occupy space left by reacted O2. (1)
(b) The brown colour would be more since the salt accelerates rusting/rust faster. (1)
17. (a) Rate increases. (1) (b) Temperature increases the kinetic energy (1) of the particles increasing the number of collisions. (1)
18. (a) N (l)
(b) R (1)
(C) M;N2 (1)
19
20. Vanadium (V) oxide is cheaper (1) and is not easily poisoned by impurities. (l)
21.
22. (a)Condensation of alcohol with higher boiling point so that it runs back to the flask as the alcohol with lower boiling point distills over. (1)
(b) Methanol. (1) It has a lower boiling point due to the size of carbon chain when compared with propanol. (1)
23. (a)Step 1 is neutralisation. (1)
(b) Step II is soda lime/ mixture of NaOH and CaO. (1)
(c) Fuel/making ethene/making hydrogen gas. (1)
24. (a)

25.

26. Natural polymers are biodegradable (1) and are expensive. (1)
Affected by acids/Not easily recyled.
27. (a) Acetone / ethanol / propanone / propanol. (1)
(b) The solvent dissolves the organic compound indicator present in the flowers / it is an organic solvent. (1)
28. (a) It absorbs carbon (IV) oxide present in the air. (1)
(b) Copper /Cuts) (1)
(c) It has rare noble gases which have not been removed / Argon. (1)
29. (a) A radical is a compound formed when elements combine to form ions / free unstable atoms or molecules / a group of free unstable atoms exist in a compound / group of atoms with a common charge. (1)
(b)
30.

4.6.2 Chemistry Paper 2 (233/2)
1 – (i)Sodium chloride / potassium chloride /rock salt.( 1 mark)
(ii) Concentrated sulphuric (VI) acid( 1 mark)
(iii) Grey solid turns green( 1 mark)
(iv)

( 1 mark)
(V)To avoid explosion.( 1 mark)
(b) (i) I The gas reacts with silver nitrate to form insoluble silver chloride.( 1 mark) II Both gases form ammonium chloride which is white.( 1 mark)
(ii)- To make hydrochloric acid.( 1 mark)
– Manufacture of ammonium chloride.( 1 mark)
– Manufacture of PVC.( 1 mark)
– Making chloroethene /vynil chloride( 1 mark)
(c)(i)imagee
(ii) Presence of Ca2+ which make water hard / forms scum. ( 1 mark)
2. (i)K – Has largest atomic radius / it most readily loses its outermost electron.( 1 mark) (ii) B/N( 1 mark)
(iii) D/Mg( 1 mark)
(iv) A (1 ) It has the smallest/smaller atomic rad1us/ its outermost electron is more/most strongly held by nucleus.(1)( 2 mark)
(v) PH is seven (7). The chlorides of group 1 elements are neutral salts.(2 marks)
(b) (i) Both CaCl2 and MgCl2 have mobile ions in molten state (1) while both CCl4 and SiCl4 are molecular compounds with no mobile ions (1).
(ii) Neon has molar mass of 20 While Flourine has a molar mass of 38 (l).
Therefore Neon diffuses faster. (l) Since it has a lower molecular mass the faster the rate of diffusion.
3
b

(d) Fuel
Solvent
Anti-septic
Manufacture of synthetic fibres
Manufacture of gasohol
Manufacture of ethanoic acid/vinegar
Used in themometers
Manufacture of other organic compounds.
Any 2 (2 marks)
4. (a) -Pressure
-Concentration
-Catalyst
-Particle size/surface area
-Light intensity (1 mark)
(b) (i)Draw a tangent to the graph at L12 min. (1) _
-Determine change in volume( Y) /Calculate gradlem
-Determine change in time.( Y)
-Divide change in volume by change in time (1)
(ii)AB Low production of gas(1) while BC the rate is very high because catalyst B was added.(1)
(iii)
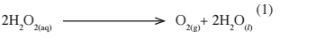
(c) (i) Lowering the temperature shifts the equilibrium to the right /favours the forward reaction. (1) Hence more S03 will be produced. (1)
(ii) Platinum or Vanadium pentoxide/vanadium (v) oxide / V2O5/ platinised asbestos any (1 mark)