2011 KCSE Chemistry Past Paper
2.3.1 Chemistry Paper 1 (233/1)
1 (a) What name is given to the process by which alcohol is formed from a carbohydrate? (1 mark)
(b) Explain why the solubility of ethane in water is lower than that of ethanol. (2 marks)
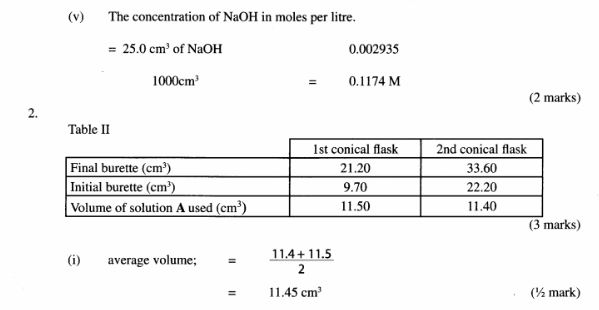
2 Complete the nuclear equation below:
(a)
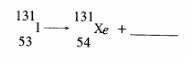
(1 mark) (b)

(c) Give one harmful effect of radioisotopes. (1 mark)
3 A mixture contains ammonium chloride, copper (II) oxide and sodium chloride.
Describe how each of the substances can be obtained from the mixture. (3 marks)
4 The set-up below shows how nitrogen gas is prepared in the laboratory.
(a) Describe how nitrogen gas is formed in the flask. (2 marks)
(b) Nitrogen is inert. State one use of the gas based on this property. (1 mark)
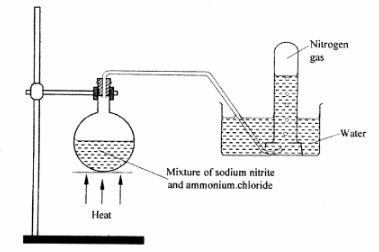
5 The diagram below represents part of the periodic table. Use it to answer the questions that follow:
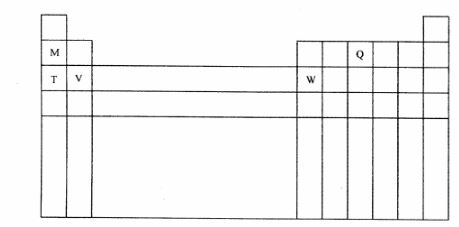
(a) Write the electronic arrangement for the stable ion formed by W. (1 mark)
(b) Write an equation for the reaction between V and Q. (1 mark)
(c) How do the ionisation energies of the elements M and T compare? Explain. (1 mark)
6 A certain mass of gas occupies 0.15dm3 at 293 K and 98,648.5Pa. Calculate its volume at 101325 Pa and 273 K. (2 marks)
7 When lead(ii) nitrate is heated. one of the products is a brown gas. (a) Write the equation of the reaction that occurs. (1 mark)
(b) If O.290dm3 of the brown gas was produced, calculate the mass of the lead(ii) nitrate that was heated. (R.F.M of lead (II) nitrate = 331; Molar gas volume = 24dm3). (2 marks)
8 (a) What is meant by a strong acid? (1 mark)
(b) In an experiment, 40cm3 of 0.5M hydrochloric acid was reacted with excess sodium carbonate and the volume of carbon (IV) oxide produced recorded with time.
In another experiment, the same volume and concentration of ethanoic acid was also reacted with excess sodium carbonate and the volume of carbon(iv) oxide produced recorded with time.
On the grid below, sketch and label the curves if the volumes of carbon (IV) oxide were plotted against time. (2 marks)
Volume of C02 cm3
9 State two reasons why hydrogen is not commonly used as a fuel. (2 marks)
10 During a class experiment, chlorine gas was bubbled into a solution of potassium iodide. (a) State the observations made. (1 mark)
(b) Using an ionic equation, explain why the reaction is redox. (2 marks)
11 Exhaust fumes of some cars contain carbon (II) oxide and other gases.
(a) Explain how carbon (II) oxide is formed in the internal combustion engines. (1 mark)
(b) Name two gases other than carbon (II) oxide that are contained in exhaust fumes and are pollutants. (2 marks)
12 Sodium hydroxide can be prepared by the following methods; I and II.
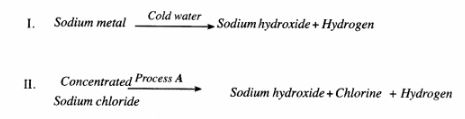
(a) Name one precaution that needs to be taken in method I. (1 mark)
(b) Give the name of process A. (1 mark)
(c) Give one use of sodium hydroxide. ( 1 mark)
13 Distinguish between the terms deliquescent and efflorescent as used in chemistry. (2 marks)
14 Two organic compounds P and Q clecolourise acidified potassium manganate (VII) solution; but only P reacts with sodium metal to give a colourless gas. Which homologous series does compound P belong? Give a reason. (2 marks)
15 Soap dissolves in water according to the equation below;
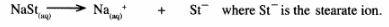
.
(a) Write the formula of the scum formed when soap is used in hard water. (1 mark)
(b) Write the ionic equation for the reaction that occurs when sodium carbonate is used to remove hardness in water. (1 mark)
16 Ethanoic acid and ethanol react as shown in the equation below:

Other than warming, how would the state of equilibrium be established within a short time? (1 mark)
17 The set up below was used to prepare a gas and study some of its properties. Study it and answer the questions that follow:
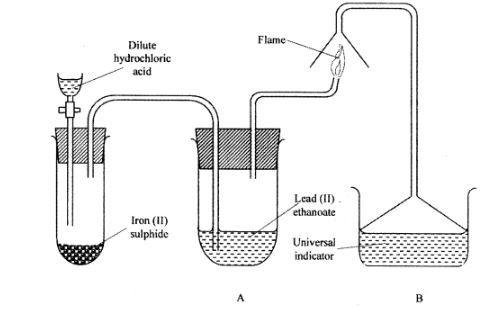
(a) State and explain the observations made in the:
(i) tube labelled A; (1 mark)
(ii) beaker labelled B. (1 mark)
(b) State one precaution that should be taken when carrying out this experiment. (1 mark)
18 Under certain conditions, chlorine gas reacts with sodium hydroxide to form sodium hypochloxite.
(a) Name the conditions under which sodium hydroxide reacts with chlorine to form sodium hypochlorite. (1 mark)
(b) State two uses of sodium hypochlorite. (2 marks)
19 50kg of ammonium sulphate (NH4)2 SO4 and 30kg of urea :CO(NH2)2 fertilizers were applied in two equal sizes of plots A and B to enrich their nitrogen content. Show by working, which plot was more enriched with nitrogen. (N = 14; S = 32; O = 16; C = 12; H = 1) (3 marks)
20 Describe how the PH of anti-acid (Actal) powder can be determined in the laboratory. (2 marks)
21 Graphite is one of the allotropes of carbon.
(a) Name one other element which exhibits allotropy. (1 mark)
22 The table below gives some properties of three elements in group (VII) of the periodic table.
Study it and answer the questions that follow:
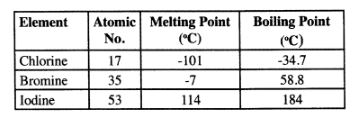
(a) Which element is in liquid fonn at room temperature? Give a reason. (1 mark)
(b) Explain why the boiling point of iodine is much higher than that of chlorine. (2 marks)
23 The thermalchemical reaction between carbon and sulphur is as shown by the equation below:
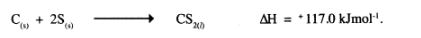
On the grid below, sketch and label the energy level diagram for the reaction. (2 marks)

24 The table below gives the number of electrons, protons and neutrons in substances X, Y and Z.
study it and answer the questions below
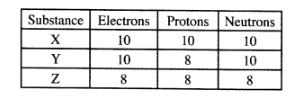
(a) Which letter represents an ion? (1 mark)
(b) Which of the substances are isotopes? Give a reason. (2 marks)
25 (a) State the Gay Lussac’s Law. (1 mark)
(b) 1Ocm3 of a gaseous hydrocarbon, C2HX required 30cm3 of oxygen for complete combustion. If steam and 20cm3 of carbon (iv) oxide were produced, what is the value of X? (2 marks)
26 The data given below was recorded when metal M was completely burnt in air. M is not the actual symbol of the metal. (R.A.M; M = 56, O =16)
Mass of empty crucible and lid = 10.240g
Mass of crucible, lid and metal M = 10.352g
Mass of crucible, lid and metal oxide = l0.400g
(a) Determine the mass of: (2 mark)
(i) metal M;
(ii) oxygen. ( 2 mark)
(b) Determine the empirical formula of me metal oxide. (2 marks)
27 The flow chart below shows some processes involved in the industrial extraction of Zinc metal
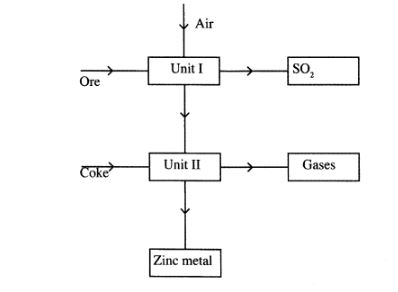
(a) Name one ore from which Zinc is extracted(1 marks)
(b) Write the equation of the reaction taking place in unit II(1 marks)
(c) Name two uses of zinc metal.(1 marks)
28 The diagram below shows the bonding between aluminium chloride and ammonia.
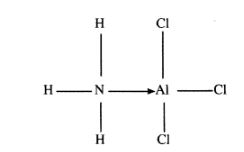
(a) Name the types of bonds that exist in the molecule.(1 marks)
(b) How many electrons are used for bonding in the molecule? (1 marks)
29. Explain why the following substances conduct an electric current.
(a) Magnesium metal.(1 marks)
(b) Molten magnesium chloride.(1 marks)
30 A sample of river water is suspectedc to contain zinc and sulphate ions.
Describe how the presence of zinc ions and sulphate ions can be established. (3 marks)
23.2 Chemistry Paper 2 (233/2)
1 The flow chart below shows some of the processes involved in large scale production of sulphuric (VI) acid. Use it to answer the questions that follow.
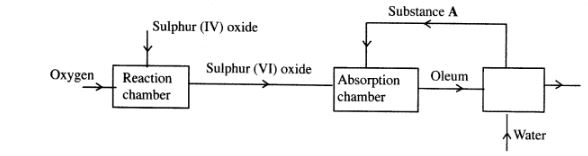
(a) Describe how oxygen is obtained from air on a large scale. (3 marks)
(b) (i) Name substance A. (1 mark)
(ii) Write an equation for the process that takes place in the absorption chamber. (1 mark)
(c) Vanadium (V) oxide is a commonly used catalyst in the contact process.
(i) Name another catalyst which can be used for this process. (1 mark)
(ii) Give two reasons why vanadium (V) oxide is the commonly used catalyst. (2 marks)
(d) State and explain the observations made when concentrated sulphuric (VI) acid is added to crystals of copper (II) sulphate in a beaker. (2 marks)
(e) The reaction of concentrated sulphuric (V1) acid with sodium chloride produces hydrogen chloride gas. State the property of concentrated sulphuric (Vi) acid illustrated in this reaction. (1 mark)
(f) Name four uses of sulphuric (VI) acid. (2 marks)
2 The set-up below was used by a student to investigate the products formed when aqueous copper (ll) chloride was electrolysed using carbon electrodes.
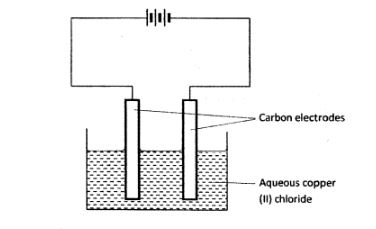
(a) (i) Write the equation for the reaction that takes place at the cathode. (1 mark) (ii) Name and describe a chemical test for the product initially formed at the anode when a highly concentrated solution of copper (ll) chloride is electrolysed. (3 marks)
(iii) How would the mass of the anode change if the carbon anode was replaced with copper metal? Explain. (2 marks)
(b) 0.6 g of metal B Were deposited when a current of O.45A was passed through an electrolyte for 72 minutes. Determine the charge on the ion of metal B. (Relative atomic mass of B = 59, 1 Faraday = 96 500 coulombs) (3 marks)
(c) The electrode potentials for cadmium and zinc are given below:
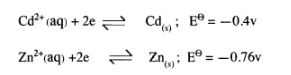
Explain why it is not advisable to store a solution of cadmium nitrate in a container made of zinc. (2 arks)
3 (a) Ethanol can be manufactured from ethene and steam as shown in the equation below:
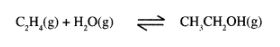
Temperature and pressure will affect the position of equilibrium of the above reaction. Name the other factor that will affect the position of equilibrium of the above reaction. (1 mark)
(b) The data in the table below was recorded when one mole of ethene was reacted with excess steam. The amount of ethanol in the equilibrium mixture was recorded under different conditions of temperature and pressure. Use the data to answer the questions that follow.
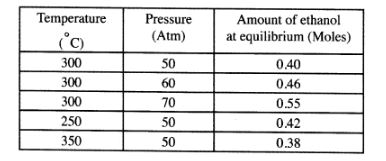
(i) State whether the reaction between ethene and steam is exothermic or endothermic. Explain your answer. (3 marks) .
(ii) State and explain one advantage and one disadvantage of using extremely high pressure in this reaction.
I. Advantage (2 marks)
II. Disadvantage (2 marks)
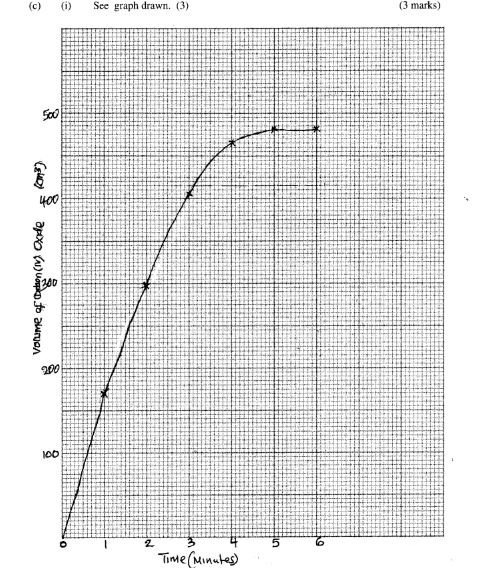
(c) In an experiment to determine the rate of reaction between calcium carbonate and dilute hydrochloric acid, 2g of calcium carbonate were reacted with excess 2 M hydrochloric acid. The volume of carbon (IV) oxide evolved was recorded at regular intervals of one minute for six minutes. The results are shown in the table below.

(i) Plot a graph of time in minutes on the horizontal axis against volume of carbon (IV) oxide on the vertical axis. (3 marks)

(ii) Determine the rate of reaction at 4 minutes. (2 marks)
4 (a) When excess calcium metal was added to 50 cm3 of 2 M aqueous copper (II) nitrate in a beaker, a brown solid and bubbles of gas were observed.
(i) Write two equations for the reactions which occurred in the beaker. (2 marks)
(ii) Explain why it is not advisable to use sodium metal for this reaction. (2 marks)
(b) Calculate the mass of calcium metal which reacted with copper (II) nitrate solution. (Relative atomic mass of Ca = 40) (2 marks)
(c) The resulting mixture in (a) above was filtered and aqueous sodium hydroxide added to the filtrate dropwise until in excess. What observations were made?(1 mark)
(d) (i) Starting with calcium oxide, describe how a solid sample of calcium carbonate can be prepared. (3 marks)
(ii) Name one use of calcium carbonate. (1 mark)
5. (a) Other than their location in the atom, name two other differences between an electron and a proton. (2 marks)
(b) The table below gives the number of electrons, protons and neutrons in particles A, B,C,D, E,F and G.
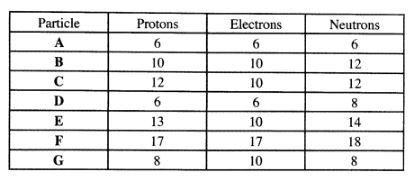
(i) Which particle is likely to be a halogen?(1 mark)
(ii) What is the mass number of E?(1 mark)
(iii) Write the formula of the compound formed when E combines with G.(1 mark)
(iv) Name the type of bond formed in (m) above.(1 mark)
(v) How does the radii of C and E compare? Give a reason.(2 marks)
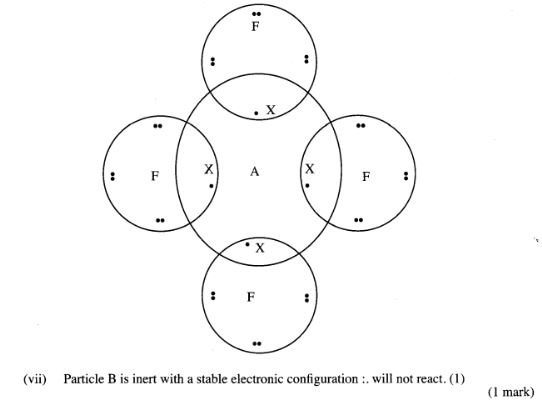
(vi) Draw a dot (.) and cross (X) diagram for the compound formed between A and F.(1 mark)
(vii) Why would particle B not react with particle D?(1 mark)
6 (a) Study the flow chart below and answer the questions that follow.
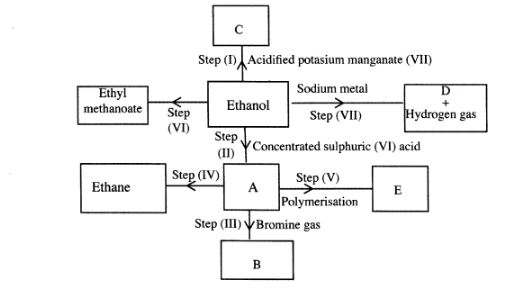
(i) I What observation will be made in Step I?(1 mark)
II Describe a chemical test that can be carried out to show the identity of compound C.(2 marks)
(ii) Give the names of the following:(2 marks)
I E …………………………. II Substance D ……………… (iii) Give the formula of substance B.(1 mark)
(iv) Name the type of reaction that occurs in:(1 mark)
I Step (II) ……………………….
II Step (IV) ……………………….
(v) Give the reagent and conditions necessary for Step (2 marks)(VI). Reagent: …………………..
Conditions ……………… ..
(b) (i) Name the following structure.
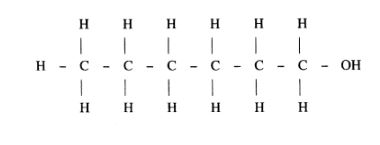
(ii) Draw the structure of an isomer of pentene. (1 mark)
7 (a) What is meant by molar heat of combustion? (1 mark)
(b) State the Hess’s Law. (1 mark)
(c) Use the following standard enthalpies of combustion of graphite, hydrogen and enthalpy of formation of propane.
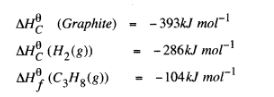
(i) Write the equation for the formation of propane. (1 mark)
(ii) Draw an energy cycle diagram that links the heat of formation of propane with its heat of combustion and the heats of combustion of graphite and hydrogen. (3 marks)
(iii) Calculate the standard heat of combustion of propane. (2 marks)
(d) Other than the enthalpy of combustion, state one factor which should be considered when choosing a fuel. (1 mark)
(e) The molar enthalpies of neutralization for dilute hydrochloric acid and dilute nitric (V)acid are -57.2k.j/mol while that of ethanoic acid is —55.2kJ/mol. Explain this observation. (2 marks)
3.3.3 Chemistry Paper 3 (233/3)
1 You are provided with:
– 1.6Og of solid A. a dibasic acid.
– Solution B containing 4.75g per litre of salt B.
– Aqueous sodium hydroxide. solution C.
– Phenolphthalein indicator.
You are required to prepare a solution of solid A and use it to determine the:-
– Concentration of sodium hydroxide. solution C
– React salt B with excess sodium hydroxide and then determine the relative molecular mass of salt B.
Procedure I
(a) Using a burette, place 25 .Ocm3 of solution B in each of two 250ml conical flasks.
Using a pipette and pipette filler, add 25.0cm3 of solution C to each of the two conical flasks. (The sodium hydroxide added is in excess). Label the conical flasks 1 and 2.
(b) Heat the contents of the first conical flask to boiling and then let the mixture boil for 5 minutes. Allow the mixture to cool.
(c) Repeat procedure (b) with the second conical flask.
While the mixtures are cooling, proceed with procedure II.
Procedure II
(a) Place all of solid A in 21 250 ml volumetric flask. Add about 150cm3 of distilled water, shake well to dissolve the solid and then add water to make up to the mark. Label this as solution A.
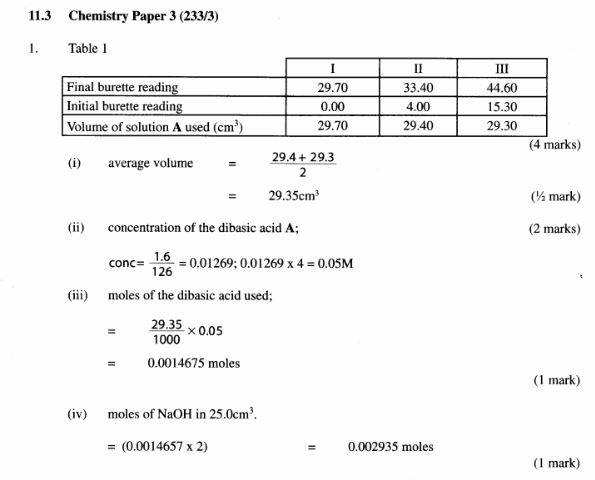
(b) Place solution A in a clean burette. Using a pipette and pipette filler, place 25 .0cm3 of solution C in a 250ml conical flask. Add 2 drops of phenolphthalein indicator and titrate with solution A. Record your results in Table 1. Repeat the titration two more times and complete the table.
Table 1
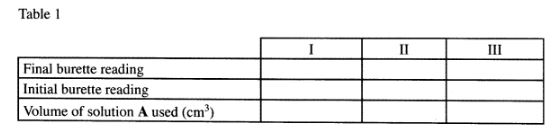
(4 marks)
Calculate the:-
(i) average volume of solution A used: (1/2 mark)
(ii) concentration in moles per litre of the dibasic acid in solution A; (2 marks) (Relative molecular mass of A is 126).
(iii) moles of the dibasic acid used; (1 mark)
(iv) moles of sodium hydroxide in 25 .0cm3 of solution C. (l mark)
(v) concentration of sodium hydroxide in moles per litre. (2 marks)
Procedure III
Add 2 drops of phenolpthalein indicator to the contents of the first conical flask prepared in procedure l and titrate with solution A. Record your results in Table 2. Repeat the procedure with the contents of the second conical flask and complete the table.
Table 2
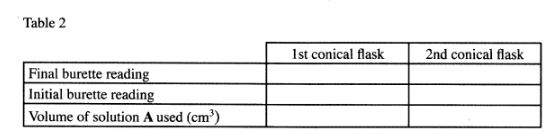
(3 marks)
Calculate the:-
(i) average volume of solution A used; (1/2 mark)
(ii) moles of the dibasic acid used; (1 mark)
(iii) moles of sodium hydroxide that reacted with the dibasic acid. (1 mark)
(iv) moles of sodium hydroxide that reacted with 25 .0cm3 of salt B in solution B; (2 marks)
(v) Given that l mole of salt B reacts with 2 moles of sodium hydroxide, calculate the: l. number of moles of salt B in 25.0cm3 of solution B; (1 mark)
ll. concentration in moles per litre of salt B in solution B; (1 mark)
lll. relative molecular mass of salt B; (2 marks)
2 (a) You are provided with solid D. Carry out the following tests and write your observations and inferences in the spaces provided.
(i) Place about one half of solid D in a test-tube and heat it strongly. Test any gases produced with both red and blue litmus papers.
Observations (2 marks)
Inferences (1 mark)
(ii) Place the rest of solid D in a boiling tube. Add about 10cm3 of distilled Water. Shake well.
To a 2cm3 portion of the solution, add about 1cm3 of hydrogen peroxide and shake well.
To the resulting mixture, add aqueous sodium hydroxide dropwise until in excess.
Observations (1 mark)
Inferences(1 mark)
(b) You are provided with solution E. Carry out the following tests and write your observations and inferences in the spaces provided.
Divide solution E into two portions.
(i) To one portion of solution E in a test-tube, add 3 drops of barium nitrate. Retain the mixture for use in test (ii) below.
Observations (1 mark)
Inferences (2 marks)
(ii) To the mixture obtained in (i) above, add about 5 cm3 of 2M nitric (V) acid.
Observations (1 mark)
Inferences (1 mark)
(iii) To portion two of solution E in a test-tube, add 2 drops of acidified potassium dichromate (VI) and warm the mixture.
3 You are provided with liquid F. Carry out the following tests and record your observations and inferences in the spaces provided.
(a) Place five drops of liquid F on a clean dry watch glass and ignite it
Observations(1 mark)
Inferences(1 mark)
(b) Place about 2cm3 of liquid F in a clean dry test tube, add all the sodium hydrogen carbonate provided.
Observations (1 mark)
Inference;(1 mark)
(c) Place about 2cm3 of liquid F in a test-tube, add about 1cm3 of acidified potassium dichromate (VI) and warm the mixture.
Observations (1 mark)
Inferences(1 mark)
2011 KCSE Chemistry-Marking Scheme/Answers